Quick Details
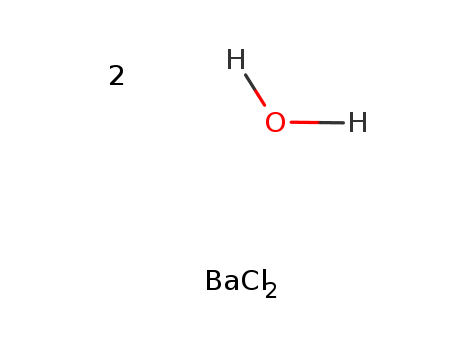
- ProName: Barium chloride
- CasNo: 10361-37-2
- Molecular Formula: BaCl2
- LimitNum: 1 Gram
Superiority
Preparation:
(1). Heat Barium Chloride Dihydrate to the temperature +150℃ and then go through the process of dehydration to obtain Barium Chloride. …
Details
Preparation:
(1). Heat Barium Chloride Dihydrate to the temperature +150℃ and then go through the process of dehydration to obtain Barium Chloride. The chemical reaction formula is as below:
BaCl2·2H2O[△]→BaCl2+2H2O2).
(2). Barium Chloride could be prepared through the following reaction routes which is usually applied in the industry scale:
BaSO4 + 4 C → BaS + 4 CO
BaS + CaCl2 → BaCl2 + CaS
Use:
Barium Chloride could used to produce other chemicals, below are the two examples:
Ba2+(aq) + SO42-(aq) → BaSO4(s)
Ba2+(aq) + C2O42-(aq) → BaC2O4(s)



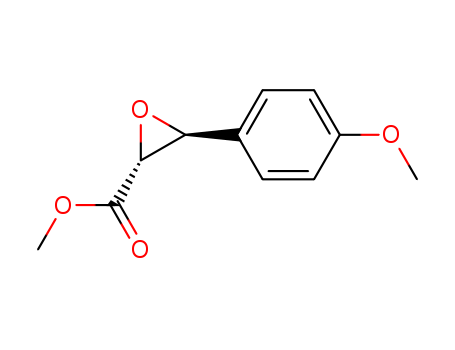
![(3aR,4R,5R,6aS)-4-[(1E,3R)-4-(3-Chlorophenoxy)-3-hydroxy-1-buten-1-yl]-5-hydroxyhexahydro-2H-cyclopenta[b]furan-2-one](http://file1.lookchem.com/300w/substances/2022-03-04-01/0de40f0a-3271-4f7d-8f67-229f602e5088.png)

![Methyl 3-[3-tert-butyl-5-(2H-benzotriazol-2-yl)-4-hydroxyphenyl]propionate](http://file1.lookchem.com/cas/reactions/2021/07/17/7724596.png)